

You can:
| Name | Somatostatin receptor type 1 |
|---|---|
| Species | Rattus norvegicus (Rat) |
| Gene | Sstr1 |
| Synonym | SRIF-2 SS-1-R SS1-R SS1R SST1 receptor |
| Disease | N/A for non-human GPCRs |
| Length | 391 |
| Amino acid sequence | MFPNGTAPSPTSSPSSSPGGCGEGVCSRGPGSGAADGMEEPGRNSSQNGTLSEGQGSAILISFIYSVVCLVGLCGNSMVIYVILRYAKMKTATNIYILNLAIADELLMLSVPFLVTSTLLRHWPFGALLCRLVLSVDAVNMFTSIYCLTVLSVDRYVAVVHPIKAARYRRPTVAKVVNLGVWVLSLLVILPIVVFSRTAANSDGTVACNMLMPEPAQRWLVGFVLYTFLMGFLLPVGAICLCYVLIIAKMRMVALKAGWQQRKRSERKITLMVMMVVMVFVICWMPFYVVQLVNVFAEQDDATVSQLSVILGYANSCANPILYGFLSDNFKRSFQRILCLSWMDNAAEEPVDYYATALKSRAYSVEDFQPENLESGGVFRNGTCASRISTL |
| UniProt | P28646 |
| Protein Data Bank | N/A |
| GPCR-HGmod model | N/A |
| 3D structure model | No available structures or models |
| BioLiP | N/A |
| Therapeutic Target Database | N/A |
| ChEMBL | CHEMBL4652 |
| IUPHAR | 355 |
| DrugBank | N/A |
| Name | CHEMBL1076623 |
|---|---|
| Molecular formula | C34H41F2N5O5S |
| IUPAC name | [(4S,4aS,8aR)-2-[(2S)-3-(2,1,3-benzothiadiazol-5-yl)-2-methylpropyl]-3,4,4a,5,6,7,8,8a-octahydro-1H-isoquinolin-4-yl]-[4-(3,4-difluorophenyl)piperazin-1-yl]methanone;(E)-but-2-enedioic acid |
| Molecular weight | 669.789 |
| Hydrogen bond acceptor | 12 |
| Hydrogen bond donor | 2 |
| XlogP | None |
| Synonyms | N/A |
| Inchi Key | IZJQLAVGJBYCQR-BDQNSCOVSA-N |
| Inchi ID | InChI=1S/C30H37F2N5OS.C4H4O4/c1-20(14-21-6-9-28-29(15-21)34-39-33-28)17-35-18-22-4-2-3-5-24(22)25(19-35)30(38)37-12-10-36(11-13-37)23-7-8-26(31)27(32)16-23;5-3(6)1-2-4(7)8/h6-9,15-16,20,22,24-25H,2-5,10-14,17-19H2,1H3;1-2H,(H,5,6)(H,7,8)/b;2-1+/t20-,22-,24-,25+;/m0./s1 |
| PubChem CID | 46880585 |
| ChEMBL | CHEMBL1076623 |
| IUPHAR | N/A |
| BindingDB | N/A |
| DrugBank | N/A |
Structure | 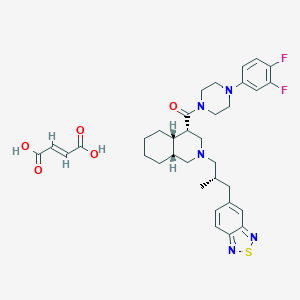 |
| Lipinski's druglikeness | This ligand has more than 10 hydrogen bond acceptor. This ligand is heavier than 500 daltons. Partition coefficient log P of this ligand is not available. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Kd | 1174.9 nM | PMID20137944 | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218