

You can:
| Name | Neuromedin-K receptor |
|---|---|
| Species | Rattus norvegicus (Rat) |
| Gene | Tacr3 |
| Synonym | Tac3r SP-N receptor Nmkr NKR NK3 receptor [ Show all ] |
| Disease | N/A for non-human GPCRs |
| Length | 452 |
| Amino acid sequence | MASVPRGENWTDGTVEVGTHTGNLSSALGVTEWLALQAGNFSSALGLPATTQAPSQVRANLTNQFVQPSWRIALWSLAYGLVVAVAVFGNLIVIWIILAHKRMRTVTNYFLVNLAFSDASVAAFNTLINFIYGLHSEWYFGANYCRFQNFFPITAVFASIYSMTAIAVDRYMAIIDPLKPRLSATATKIVIGSIWILAFLLAFPQCLYSKIKVMPGRTLCYVQWPEGPKQHFTYHIIVIILVYCFPLLIMGVTYTIVGITLWGGEIPGDTCDKYHEQLKAKRKVVKMMIIVVVTFAICWLPYHVYFILTAIYQQLNRWKYIQQVYLASFWLAMSSTMYNPIIYCCLNKRFRAGFKRAFRWCPFIQVSSYDELELKTTRFHPTRQSSLYTVSRMESVTVLFDPNDGDPTKSSRKKRAVPRDPSANGCSHRGSKSASTTSSFISSPYTSVDEYS |
| UniProt | P16177 |
| Protein Data Bank | N/A |
| GPCR-HGmod model | N/A |
| 3D structure model | No available structures or models |
| BioLiP | N/A |
| Therapeutic Target Database | N/A |
| ChEMBL | CHEMBL3154 |
| IUPHAR | 362 |
| DrugBank | N/A |
| Name | Talnetant |
|---|---|
| Molecular formula | C25H22N2O2 |
| IUPAC name | 3-hydroxy-2-phenyl-N-[(1S)-1-phenylpropyl]quinoline-4-carboxamide |
| Molecular weight | 382.463 |
| Hydrogen bond acceptor | 3 |
| Hydrogen bond donor | 2 |
| XlogP | 5.7 |
| Synonyms | (-)-3-Hydroxy-2-phenyl-N-[1(S)-phenylpropyl]quinoline-4-carboxamide 3-hydroxy-2-phenyl-N-(1-phenylpropyl)quinoline-4-carboxamide 4080AH CHEMBL10188 HY-14552 [ Show all ] |
| Inchi Key | BIAVGWDGIJKWRM-FQEVSTJZSA-N |
| Inchi ID | InChI=1S/C25H22N2O2/c1-2-20(17-11-5-3-6-12-17)27-25(29)22-19-15-9-10-16-21(19)26-23(24(22)28)18-13-7-4-8-14-18/h3-16,20,28H,2H2,1H3,(H,27,29)/t20-/m0/s1 |
| PubChem CID | 5311424 |
| ChEMBL | CHEMBL10188 |
| IUPHAR | 2132 |
| BindingDB | 50051293 |
| DrugBank | N/A |
Structure | 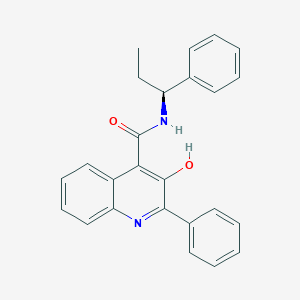 |
| Lipinski's druglikeness | This ligand has a partition coefficient log P greater than 5. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Ki | 39.8107 - 50.1187 nM | PMID11226387 | IUPHAR |
| Ki | 190.0 nM | PMID21376585 | BindingDB,ChEMBL |
| Ki | 193.0 nM | PMID25738882 | BindingDB,ChEMBL |
| Ki | 199.53 nM | PMID25738882 | ChEMBL |
| Ki | 200.0 nM | PMID25738882 | BindingDB |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218