

You can:
| Name | Glucagon-like peptide 1 receptor |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | GLP1R |
| Synonym | glucagon-like peptide 1 receptor GLP-1R GLP-1-R GLP-1 receptor |
| Disease | Type 1/2 diabetes Type 1 diabetes Obesity Non-insulin dependent diabetes Non-alcoholic steatohepatitis [ Show all ] |
| Length | 463 |
| Amino acid sequence | MAGAPGPLRLALLLLGMVGRAGPRPQGATVSLWETVQKWREYRRQCQRSLTEDPPPATDLFCNRTFDEYACWPDGEPGSFVNVSCPWYLPWASSVPQGHVYRFCTAEGLWLQKDNSSLPWRDLSECEESKRGERSSPEEQLLFLYIIYTVGYALSFSALVIASAILLGFRHLHCTRNYIHLNLFASFILRALSVFIKDAALKWMYSTAAQQHQWDGLLSYQDSLSCRLVFLLMQYCVAANYYWLLVEGVYLYTLLAFSVLSEQWIFRLYVSIGWGVPLLFVVPWGIVKYLYEDEGCWTRNSNMNYWLIIRLPILFAIGVNFLIFVRVICIVVSKLKANLMCKTDIKCRLAKSTLTLIPLLGTHEVIFAFVMDEHARGTLRFIKLFTELSFTSFQGLMVAILYCFVNNEVQLEFRKSWERWRLEHLHIQRDSSMKPLKCPTSSLSSGATAGSSMYTATCQASCS |
| UniProt | P43220 |
| Protein Data Bank | 5vex, 3c59, 3c5t, 5nx2, 3iol, 4zgm, 5otu, 5vew, 5otw, 5otx, 5otv |
| GPCR-HGmod model | P43220 |
| 3D structure model | This structure is from PDB ID 5vex. |
| BioLiP | BL0418498,BL0418499, BL0143794, BL0143795, BL0167479, BL0167480, BL0324354, BL0324355,BL0324356, BL0378791,BL0378792, BL0379513,BL0379514, BL0418500,BL0418501, BL0380967, BL0418494,BL0418495, BL0418496,BL0418497, BL0143732, BL0143731, BL0380966 |
| Therapeutic Target Database | T36075 |
| ChEMBL | CHEMBL1784 |
| IUPHAR | 249 |
| DrugBank | BE0000857 |
| Name | MLS001362111 |
|---|---|
| Molecular formula | C28H37F3N2O6S |
| IUPAC name | methyl (3aR,4S,9aS,9bS)-2-ethyl-4-(4-hexylsulfanylphenyl)-1,3-dioxo-4,6,7,8,9,9b-hexahydro-3aH-pyrrolo[3,4-a]indolizine-9a-carboxylate;2,2,2-trifluoroacetic acid |
| Molecular weight | 586.667 |
| Hydrogen bond acceptor | 11 |
| Hydrogen bond donor | 1 |
| XlogP | None |
| Synonyms | HMS2199P05 CHEMBL3213970 SMR001217130 |
| Inchi Key | AAJBGBSJHNWVDG-CPNWZACFSA-N |
| Inchi ID | InChI=1S/C26H36N2O4S.C2HF3O2/c1-4-6-7-10-17-33-19-13-11-18(12-14-19)22-20-21(24(30)27(5-2)23(20)29)26(25(31)32-3)15-8-9-16-28(22)26;3-2(4,5)1(6)7/h11-14,20-22H,4-10,15-17H2,1-3H3;(H,6,7)/t20-,21-,22-,26+;/m1./s1 |
| PubChem CID | 53312927 |
| ChEMBL | CHEMBL3213970 |
| IUPHAR | N/A |
| BindingDB | N/A |
| DrugBank | N/A |
Structure | 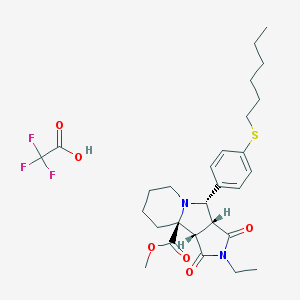 |
| Lipinski's druglikeness | This ligand has more than 10 hydrogen bond acceptor. This ligand is heavier than 500 daltons. Partition coefficient log P of this ligand is not available. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Potency | 5623.4 nM | PubChem BioAssay data set | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218