

You can:
| Name | Proteinase-activated receptor 1 |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | F2R |
| Synonym | Coagulation factor II receptor PAR-1 coagulation factor II (thrombin) receptor protease-activated receptor 1 Thrombin receptor [ Show all ] |
| Disease | Thrombosis Restenosis Myocardial infarction Cancer Atherosclerosis [ Show all ] |
| Length | 425 |
| Amino acid sequence | MGPRRLLLVAACFSLCGPLLSARTRARRPESKATNATLDPRSFLLRNPNDKYEPFWEDEEKNESGLTEYRLVSINKSSPLQKQLPAFISEDASGYLTSSWLTLFVPSVYTGVFVVSLPLNIMAIVVFILKMKVKKPAVVYMLHLATADVLFVSVLPFKISYYFSGSDWQFGSELCRFVTAAFYCNMYASILLMTVISIDRFLAVVYPMQSLSWRTLGRASFTCLAIWALAIAGVVPLLLKEQTIQVPGLNITTCHDVLNETLLEGYYAYYFSAFSAVFFFVPLIISTVCYVSIIRCLSSSAVANRSKKSRALFLSAAVFCIFIICFGPTNVLLIAHYSFLSHTSTTEAAYFAYLLCVCVSSISCCIDPLIYYYASSECQRYVYSILCCKESSDPSSYNSSGQLMASKMDTCSSNLNNSIYKKLLT |
| UniProt | P25116 |
| Protein Data Bank | 3vw7 |
| GPCR-HGmod model | P25116 |
| 3D structure model | This structure is from PDB ID 3vw7. |
| BioLiP | BL0217099 |
| Therapeutic Target Database | T36483 |
| ChEMBL | CHEMBL3974 |
| IUPHAR | 347 |
| DrugBank | BE0000928 |
| Name | CHEMBL3091993 |
|---|---|
| Molecular formula | C24H27FN2O2 |
| IUPAC name | [(1R,3aS,7aS)-1-[(E)-2-[5-(3-fluorophenyl)pyridin-2-yl]ethenyl]-2-methyl-1,3,3a,4,5,6,7,7a-octahydroinden-2-yl] carbamate |
| Molecular weight | 394.49 |
| Hydrogen bond acceptor | 4 |
| Hydrogen bond donor | 1 |
| XlogP | 5.5 |
| Synonyms | BDBM50443580 |
| Inchi Key | BUPBXZWQINIBSW-QKASLZCDSA-N |
| Inchi ID | InChI=1S/C24H27FN2O2/c1-24(29-23(26)28)14-17-5-2-3-8-21(17)22(24)12-11-20-10-9-18(15-27-20)16-6-4-7-19(25)13-16/h4,6-7,9-13,15,17,21-22H,2-3,5,8,14H2,1H3,(H2,26,28)/b12-11+/t17-,21-,22+,24?/m0/s1 |
| PubChem CID | 72547556 |
| ChEMBL | CHEMBL3091993 |
| IUPHAR | N/A |
| BindingDB | N/A |
| DrugBank | N/A |
Structure | 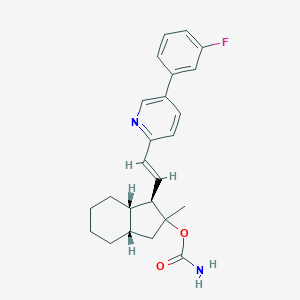 |
| Lipinski's druglikeness | This ligand has a partition coefficient log P greater than 5. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| IC50 | 2.7 nM | PMID24900604 | ChEMBL |
| IC50 | 2.9 nM | PMID24900604 | ChEMBL |
| IC50 | 100.0 nM | PMID24900604 | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218