

You can:
| Name | Melanocortin receptor 4 |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | MC4R |
| Synonym | MC4-R MC4 receptor |
| Disease | Obesity; Sexual dysfunction Obesity; Diabetes Obesity Metabolic disorders Sexual dysfunction [ Show all ] |
| Length | 332 |
| Amino acid sequence | MVNSTHRGMHTSLHLWNRSSYRLHSNASESLGKGYSDGGCYEQLFVSPEVFVTLGVISLLENILVIVAIAKNKNLHSPMYFFICSLAVADMLVSVSNGSETIVITLLNSTDTDAQSFTVNIDNVIDSVICSSLLASICSLLSIAVDRYFTIFYALQYHNIMTVKRVGIIISCIWAACTVSGILFIIYSDSSAVIICLITMFFTMLALMASLYVHMFLMARLHIKRIAVLPGTGAIRQGANMKGAITLTILIGVFVVCWAPFFLHLIFYISCPQNPYCVCFMSHFNLYLILIMCNSIIDPLIYALRSQELRKTFKEIICCYPLGGLCDLSSRY |
| UniProt | P32245 |
| Protein Data Bank | N/A |
| GPCR-HGmod model | P32245 |
| 3D structure model | This predicted structure model is from GPCR-EXP P32245. |
| BioLiP | N/A |
| Therapeutic Target Database | T72458 |
| ChEMBL | CHEMBL259 |
| IUPHAR | 285 |
| DrugBank | N/A |
| Name | D0I8SJ |
|---|---|
| Molecular formula | C90H127N25O26S |
| IUPAC name | (4S)-5-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[2-[[(2S)-6-amino-1-[(2S)-2-[[(2S)-1-(carboxymethylamino)-3-methyl-1-oxobutan-2-yl]carbamoyl]pyrrolidin-1-yl]-1-oxohexan-2-yl]amino]-2-oxoethyl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-3-(1H-imidazol-4-yl)-1-oxopropan-2-yl]amino]-4-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-1-[(2S)-3-carboxy-2-[[(2S)-2,4-diamino-4-oxobutanoyl]amino]propanoyl]pyrrolidine-2-carbonyl]amino]-3-hydroxypropanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-3-hydroxypropanoyl]amino]-4-methylsulfanylbutanoyl]amino]-5-oxopentanoic acid |
| Molecular weight | 2007.21 |
| Hydrogen bond acceptor | 31 |
| Hydrogen bond donor | 27 |
| XlogP | -10.1 |
| Synonyms | N/A |
| Inchi Key | BUVGQEXSQXRZQG-LBQSHGEUSA-N |
| Inchi ID | InChI=1S/C90H127N25O26S/c1-47(2)74(87(139)100-43-73(125)126)113-86(138)68-21-13-31-114(68)88(140)59(18-9-10-29-91)102-70(120)42-99-76(128)62(36-50-40-98-55-17-8-7-16-53(50)55)108-77(129)56(19-11-30-97-90(94)95)103-80(132)60(34-48-14-5-4-6-15-48)106-82(134)63(37-51-41-96-46-101-51)109-78(130)57(26-27-71(121)122)104-79(131)58(28-33-142-3)105-83(135)65(44-116)111-81(133)61(35-49-22-24-52(118)25-23-49)107-84(136)66(45-117)112-85(137)67-20-12-32-115(67)89(141)64(39-72(123)124)110-75(127)54(92)38-69(93)119/h4-8,14-17,22-25,40-41,46-47,54,56-68,74,98,116-118H,9-13,18-21,26-39,42-45,91-92H2,1-3H3,(H2,93,119)(H,96,101)(H,99,128)(H,100,139)(H,102,120)(H,103,132)(H,104,131)(H,105,135)(H,106,134)(H,107,136)(H,108,129)(H,109,130)(H,110,127)(H,111,133)(H,112,137)(H,113,138)(H,121,122)(H,123,124)(H,125,126)(H4,94,95,97)/t54-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,74-/m0/s1 |
| PubChem CID | 44400194 |
| ChEMBL | CHEMBL415165 |
| IUPHAR | N/A |
| BindingDB | 50165931 |
| DrugBank | N/A |
Structure | 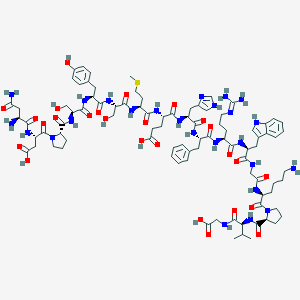 |
| Lipinski's druglikeness | This ligand has more than 5 hydrogen bond donor. This ligand has more than 10 hydrogen bond acceptor. This ligand is heavier than 500 daltons. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| EC50 | 0.19 nM | PMID15857110 | BindingDB |
| EC50 | 0.19 nM | PMID15857110 | ChEMBL |
| EC50 | 0.36 nM | PMID15857110 | ChEMBL |
| EC50 | 0.36 nM | PMID15857110 | BindingDB |
| Ki | 0.31 nM | PMID15857110 | ChEMBL |
| Ki | 0.31 nM | PMID15857110 | BindingDB |
| Relative efficacy | 100.0 % | PMID15857110 | ChEMBL |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218