

You can:
| Name | SCHEMBL13207001 |
|---|---|
| Molecular formula | C19H23N5O4S |
| IUPAC name | 2-hydroxy-N,N-dimethyl-3-[[4-[[(1R)-1-(5-methylfuran-2-yl)propyl]amino]-1-oxo-1,2,5-thiadiazol-3-yl]amino]benzamide |
| Molecular weight | 417.484 |
| Hydrogen bond acceptor | 7 |
| Hydrogen bond donor | 3 |
| XlogP | 2.5 |
| Synonyms | BDBM50226999 2-hydroxy-N,N-dimethyl-3-{4-[(R)-1-(5-methyl-furan-2-yl)-propylamino]-1-oxo-1H-1lambda4-[1,2,5]thiadiazol-3-ylamino}-benzamide |
| Inchi Key | BGHMYZUFTBKLKU-RNHBAAACSA-N |
| Inchi ID | InChI=1S/C19H23N5O4S/c1-5-13(15-10-9-11(2)28-15)20-17-18(23-29(27)22-17)21-14-8-6-7-12(16(14)25)19(26)24(3)4/h6-10,13,25H,5H2,1-4H3,(H,20,22)(H,21,23)/t13-,29?/m1/s1 |
| PubChem CID | 136097219 |
| ChEMBL | CHEMBL253306 |
| IUPHAR | N/A |
| BindingDB | 50226999 |
| DrugBank | N/A |
Structure | 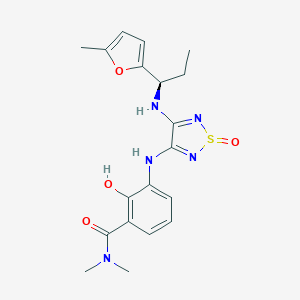 |
| Lipinski's druglikeness | This ligand satisfies Lipinski's rule of five. |
You can:
| GLASS ID | Name | UniProt | Gene | Species | Length |
|---|---|---|---|---|---|
| 557989 | C-X-C chemokine receptor type 1 | P25024 | CXCR1 | Homo sapiens (Human) | 350 |
| 557990 | C-X-C chemokine receptor type 2 | P25025 | CXCR2 | Homo sapiens (Human) | 360 |
zhanglab![]() zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218
zhanggroup.org | (734) 647-1549 | 100 Washtenaw Avenue, Ann Arbor, MI 48109-2218